
I was reading the labels on my shampoo today and toothpaste today, and I noticed that the toothpaste had sodium lauryl sulfate as an ingredient, while the shampoo had sodium laureth sulfate. I initially assumed the two were corporate-speak for the same thing, but to my surprise, they actually were not.


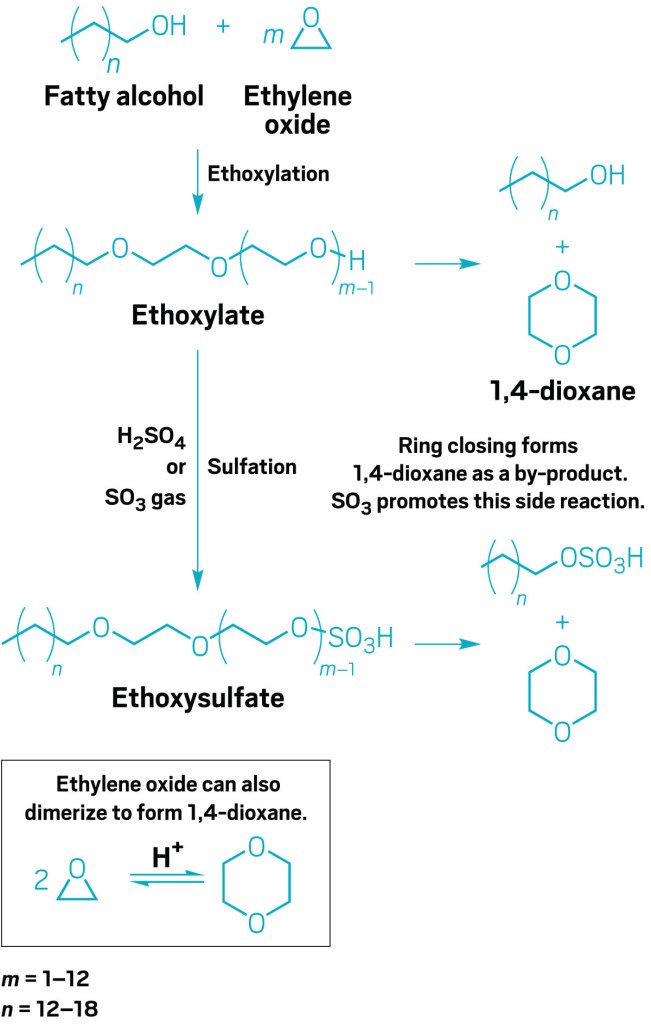
Both sodium lauryl sulfate (SLS) and sodium laureth sulfate (SLES) are anionic detergents found in a variety of cleaning products, including soaps, shampoos, and toothpaste. SLS is produced by treating lauryl alcohol (a hydrogenated derivative of coconut/ palm oil) with sulfur trioxide gas, oleum, or chlorosulfuric acid. Of note, lauryl alcohol in the commercial form actually consists of a variety of chain lengths, so SLS in soaps is actually a mixture of sodium alkyl sulfates. By contrast, SLES is created by ethoxylation of lauryl alcohol, which is then converted to a half ester of sulfuric acid.
Confusingly, both SLS and SLES go by other names. Sodium lauryl sulfate is also known as sodium dodecyl sulfate (SDS), of electrophoresis fame (discussed more below). Sodium laureth sulfate is a contraction of sodium lauryl ether sulfate (which certainly lends more clarity to the structure).
Functionally, SLES is less abrasive and has better foaming characteristics compared to SLS. However, 1,4-dioxane, a possible carcinogen, is also a by-product of the ethoxylation step of SLES production. Although no federal regulations require the removal of 1,4-dioxane, New York state has recently passed laws decreasing the acceptable concentration of 1,4-dioxane in cleaning products to 1 ppm by 2023. California is considering something similar.
Other Applications
SLS has a variety of other applications including as a food additive that aids in emulsification, an ionic stabilizer in creams/lotions, and as a medium to suspend carbon nanotubes.
Perhaps the application most familiar to the biology student is SDS-PAGE. Complex mixtures of proteins are dissolved in a solution that includes SLS/SDS. SLS is an excellent detergent that unfolds proteins, rendering all proteins a similar shape. Moreover, the amount of SLS bound to each protein is proportional to the length of the protein. SLS-solubilized proteins are then run through a slab of polyacrylamide gel subjected to an electric field. Larger proteins move slower through the gel than smaller proteins, allowing for separation by size.
SDS is also used in CLARITY, a method of imaging neuronal tissues. In its original formulation in 2013, a tissue sample is first prepared into a nanoporous hydrogel-tissue hybrid which preserves the location of proteins and nucleic acids. This is then rendered transparent by dissolving lipids in detergents such as SLS. The process is quite time-consuming but yields gorgeous images!
Not at all related to SLS, but on the subject of Prof. Deisseroth (who I am certain will win a Nobel Prize within the next decade)- a slightly older, but fascinating article from StatNews about the origins of optogenetics.
References
- Chung K, Wallace J, Kim SY, et al. Structural and molecular interrogation of intact biological systems. Nature. 2013;497(7449):332-337. doi:10.1038/nature12107
- https://en.wikipedia.org/wiki/Sodium_dodecyl_sulfate
- https://en.wikipedia.org/wiki/Sodium_laureth_sulfate
- https://cen.acs.org/business/consumer-products/companies-getting-14-dioxane-home/98/i11
- Alberts, B. (2015). Molecular biology of the cell. Page 452-455.
- https://www.statnews.com/2016/09/01/optogenetics/
- https://directorsblog.nih.gov/2013/04/11/the-brain-now-you-see-it-soon-you-wont